Privilege Check: Cis vs Trans Fats
*Technically we’re talking about cis and trans fatty acids, but the more common terminology is cis and trans fat.
Moral of the story: trans fats are bad for our health & unrefined oils > butter >>>>>>>>>>> margarine/shortening/hydrogenated oils.
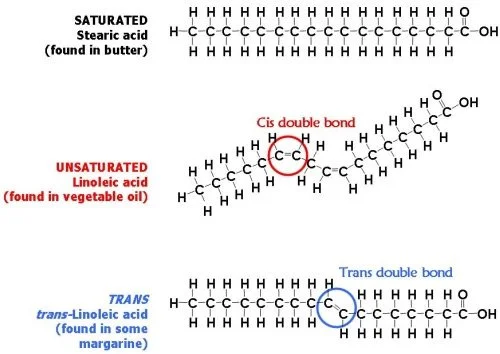
What's the difference between cis and trans fats?
Diagram by Vincent van Ginneken
Both cis and trans fats are unsaturated, meaning that there's at least one double bond in the chain. The double bond in trans fats makes them linear, and harder to break down in your body. A fat molecule is monounsaturated if it contains one double bond, and polyunsaturated if it contains more than one double bond. I could throw in some organic chemistry jargon here, but what's important to note is that cis fats are generally kinked, and trans fats are linear. This plays a role in membrane fluidity, which will be discussed later (spoiler: we want our fats to be kinked and cell membranes to be fluid). Monounsaturated fats (e.g. olive oil, avocados, nuts) and polyunsaturated fats (e.g. salmon, various veggie oils) oils are good for you. Saturated and trans fats? Not so much.
WHY DO WE CARE ABOUT TRANS FATS?
Companies use trans fats because they're cheap to manufacture, increase the shelf-life of products, and improve the taste & texture of foods. The American Heart Association states that trans fats can increase bad LDL cholesterol and decrease healthy HDL cholesterol levels, leading to a higher risk of cardiovascular disease. That's no bueno because heart disease is the leading cause of death for people of most ethnicities in the US. We rely on our hearts to circulate blood around our bodies, and they have a harder time doing that if our arteries are lined with LDL plaque. On the other hand, HDL helps rid your body of excess cholesterol so it's less likely to end up in your arteries. (yay for oatmeal and its LDLC-lowering capabilities)
WHAT FOODS HAVE TRANS FATS?
The AHA recommends consuming less than 1 percent of your calories from trans fatty acids. Which basically means, stay as far as you can from trans fats as possible. That's pretty hard, though, considering you're likely to find them in fried foods (e.g. doughnuts), margarines, shortenings, and many baked goods (e.g. cookies, pies, crackers, pizza dough).
In 2003 the Food and Drug Administration (FDA) issued a regulation on trans fats, allowing manufacturers to place no more than 0.5 grams of trans fats, per serving, in any given food. This has been controversial, as many doctors and scientists agree that 0.5 grams per serving is much too lenient. You can determine the amount of trans fats in a particular packaged food by looking at the Nutrition Facts panel. The sneaky part is that products can be listed as “0 grams of trans fats” if they contain anywhere between 0-0.5 g of trans fat per serving. Anything that mentions “partially hydrogenated oils" contains trans fats.
WHAT MAKES TRANS FATS (AND SATURATED FATS) SO UNHEALTHY?
Dan S, who has 744 points in the chemistry stack exchange community, breaks it down for us:
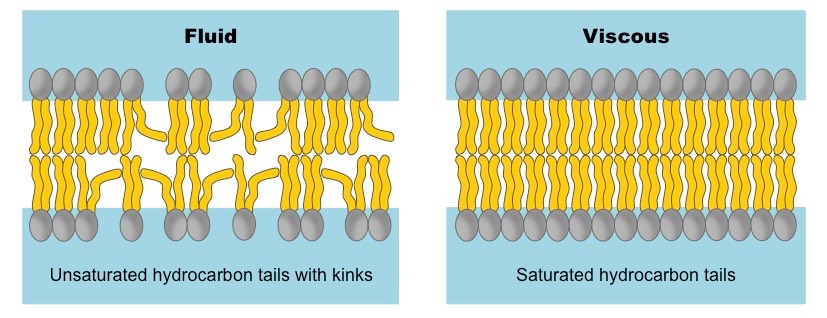
left photo is good. our cells consist of phospholipid bilayers. kinks in the tails of these phospholipids maintain a level of membrane fluidity that facilitates cell-to-cell signalling. right photo is bad. (source: BioNinja)
To put it simply, the more straight fatty acids in the membrane, the more rigid (i.e. less fluid) it will be. The more "interruptions" to the hydrophobic interactions between the fatty acids' tails (by "kinks" in the tails or cholesterol), the more fluid the membrane will be. The above picture shows two distinct states of the membrane.
On the right-hand side the membrane contains mostly straight (i.e. saturated and/or trans) fatty acids, which results in a more rigid and viscous membrane. This rigid state makes it more difficult for the cell to communicate with its surroundings (chemical signals cannot enter the cell easily, proteins in the membrane are less mobile, etc.).
On the left-hand side, the membrane contains more unsaturated fatty acids of the cis conformation. The "kink" that characterizes cis-fatty acids is the main reason this membrane is more fluid - it makes it harder for the fatty acids' tails to stick together due to hydrophobic attraction. Cholesterol serves the same purpose as those "kinks", by disrupting the attraction between fatty acids' hydrophobic tails. This is a very important role of cholesterol in virtually all of the cells in the body, which makes cholesterol a very important molecule in the body.
tl;dr: "Trans-fatty acids are considered unhealthy because when consumed, they will mostly remain intact (i.e. will not be metabolized; as this form of fatty acids is not found in nature, the body's enzymes have a limited ability to catabolize them), and then they can penetrate cellular membranes, form rigid hydrophobic bonds with the other fatty acids, and decrease the membrane's overall fluidity.
Are there naturally occurring trans fats?
AHA: "Small amounts of trans fats occur naturally in some meat and dairy products, including beef, lamb and butterfat. There have not been sufficient studies to determine whether these naturally occurring trans fats have the same bad effects on cholesterol levels as trans fats that have been industrially manufactured."
Should I use butter or margarine?
Previously, margarine was thought to be healthier than butter because margarine has less saturated fat. However, the process of hydrogenating vegetable oil (i.e. literally adding hydrogen atoms) to make it into margarine creates trans fats, which butter doesn't have. Generally, the more solid the margarine, the higher its trans fat content is. If you can’t switch to buying butter (Kerrygold’s Irish grass fed butter is my ultimate fave and yes, i'm enough of a foodie to have a favorite kind of butter), look for margarines/alternative spreads that are lower in saturated fat, higher in unsaturated fat, and hopefully free of trans fat.
What about cooking with butter vs olive oil?
Fats heated beyond their smoke point give off carcinogenic smoke. Virgin olive oil has a smoke point of 325-375 deg F. Butter has a smoke point of 350 deg F. Refined olive oil has a smoke point of 465+ deg F. Clarified butter, or ghee, which has most of the milk proteins removed, has a higher smoke point (485 deg F). I don't usually have ghee on hand, so I cook with refined olive oil or coconut oil (which has a smoke point of 350 deg F). However, I use butter as called for in baking recipes and as a finishing touch for dishes (most often for steaks & corn) because it's more flavorful than extra virgin olive oil (EVOO), and I never use margarine or shortening (e.g. Crisco). More about various cooking fats and their smoke points on Serious Eats.